Evaluation and Management of Delirium
Purpose
Provide guidance on the evaluation, diagnosis, and management of hospitalized patients who develop delirium.
Background/Definitions
Delirium is a neuropsychiatric disorder that is characterized by a disturbance in attention, consciousness and cognition with a reduced ability to focus, sustain or shift attention. It can develop over a short period of time, is a change from baseline, and fluctuates in severity. The clinical presentation varies but usually presents with psychomotor behavioral disturbances such as hyperactivity or hypoactivity and with impairment in sleep duration and quality.
Delirium is caused by an underlying medical condition that is not better explained by another preexisting, evolving, or established neurocognitive disorder. The underlying cause of delirium can vary widely and involve anything that stresses the baseline homeostasis of a vulnerable patient. Examples include: substance abuse intoxication and withdrawal, medication side effects, infection, surgery, metabolic derangements, pain, constipation, and urinary retention.
There are 3 subtypes of delirium:
- Hyperactive: patients present with restlessness, purposeless and uncontrollable movements, agitation, hallucinations, and behaviors
- Hypoactive: patients appear calm, lethargic, and have slowed mentation and slow/decreased movements.
- Mixed: fluctuation between hyperactive and hypoactive states.
Delirium has consistently shown to be associated with higher mortality rates, longer ICU and hospital lengths of stay, increased morbidity, and cognitive and psychiatric sequelae that can persist weeks to months following hospital discharge.
The elderly, polytrauma patients and those critically ill in the ICU are all groups that have been identified as particularly susceptible to developing delirium. The incidence of delirium in trauma patients admitted to the ICU has been reported as up to 67%, with increased risk for elderly and those requiring mechanical ventilation.
In light of this, it is critical for trauma and critical care providers to be well versed in screening for and identifying delirium as well as implementing preventative strategies against delirium in order to optimize patient outcomes and reduce healthcare costs.
Guideline Inclusion Criteria
All admitted trauma patients
Guideline Exclusion Criteria
none
Diagnostic Evaluation
· Risk factors for delirium development:
Each trauma patient should be assessed for nonmodifiable and modifiable risk factors that may contribute to the development of delirium.
|
Nonmodifiable Risk Factors |
Modifiable Risk Factors |
|
Increased age |
Restraints |
|
Depressed GCS on arrival |
Ventilator days |
|
Increased blood product transfusion |
Increased sedation |
|
Multisystem organ failure |
Infection/sepsis |
|
Traumatic brain injury (TBI) |
Indwelling urinary catheters/lines |
|
History of substance abuse |
Medications |
|
Frailty |
|
|
Comorbidities (hypertension, dementia) |
|
|
Nutritional impairment |
|
|
Medications known to be associated with increased delirium can include: |
||||||||||||||||
|
· Patient care should be centered around optimizing modifiable risk factors as able in hopes of minimizing the risk of delirium development.
· Delirium Detection and monitoring:
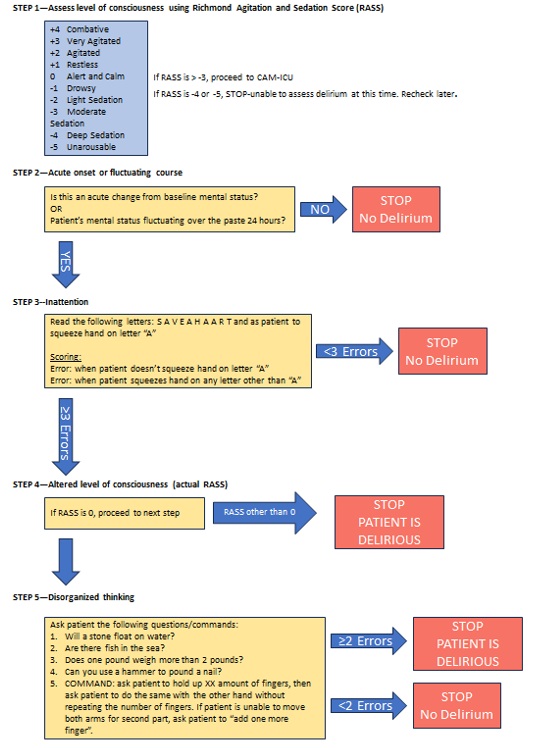
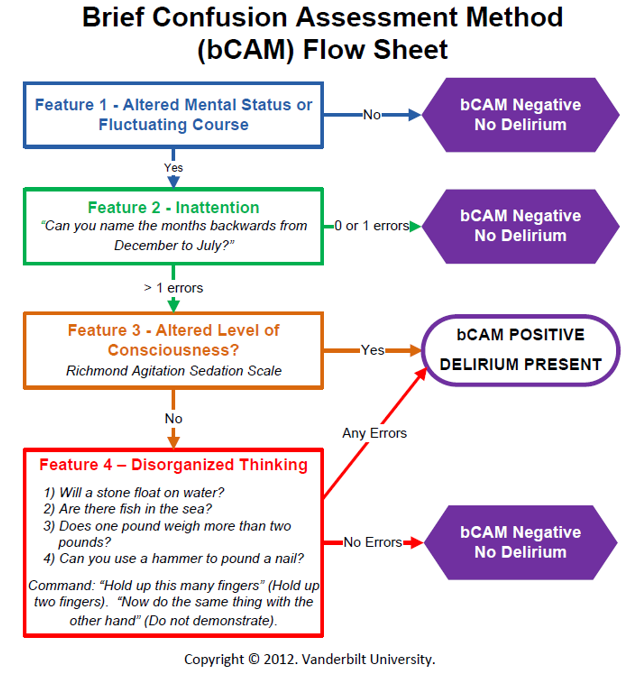
The most reliable method for detecting delirium is with the use of externally validated screening tools. One of the more widely used screening tool is the Confusion Assessment Method for ICU (CAM-ICU) which is applied primarily to patients in the ICU. Alternatively, a Brief Confusion Assessment Method (bCAM) is primarily used for delirium screening on floor patients. (see Figure 1)
Practice Recommendations for Management
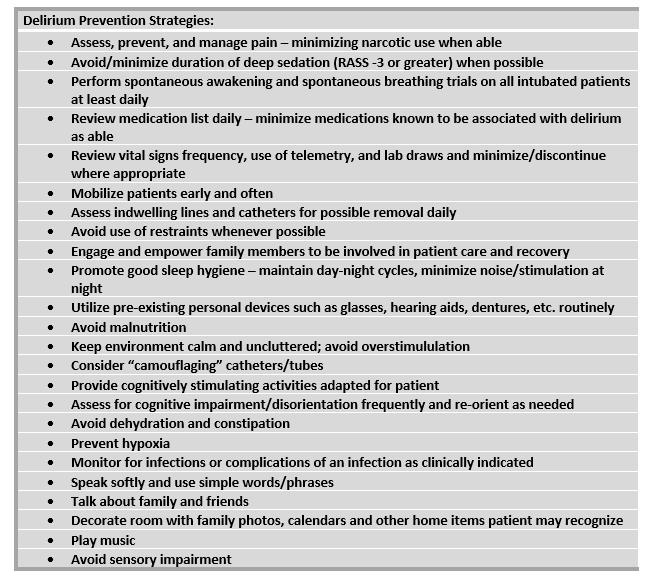
- All trauma patients should be assessed for delirium risk and development daily and as needed when a change in clinical status occurs and delirium is suspected. This includes optimizing modifiable risk factors and medication regimens as able and implementing delirium prevention strategies. (see Figure 2)
- If delirium is suspected, diagnosis should be confirmed by using the CAM-ICU or bCAM screening tools.
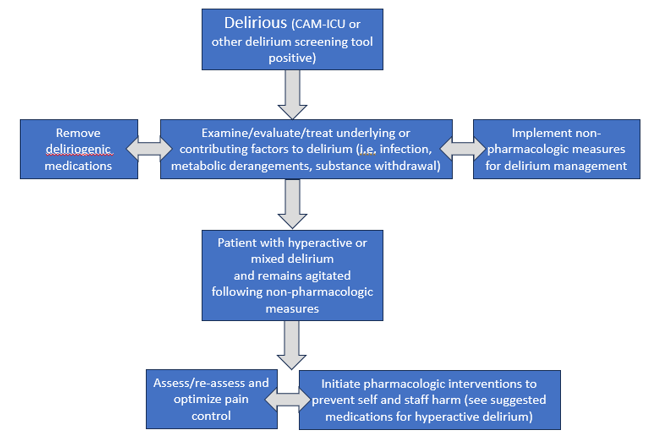
- Once delirium is confirmed, provider should examine/evaluate the patient for possible causes and contributing factors to delirium (i.e. infection, electrolyte abnormalities, medication side effects, substance intoxication/withdrawal, etc) and treat/correct as indicated by work-up (See FIGURE 3).
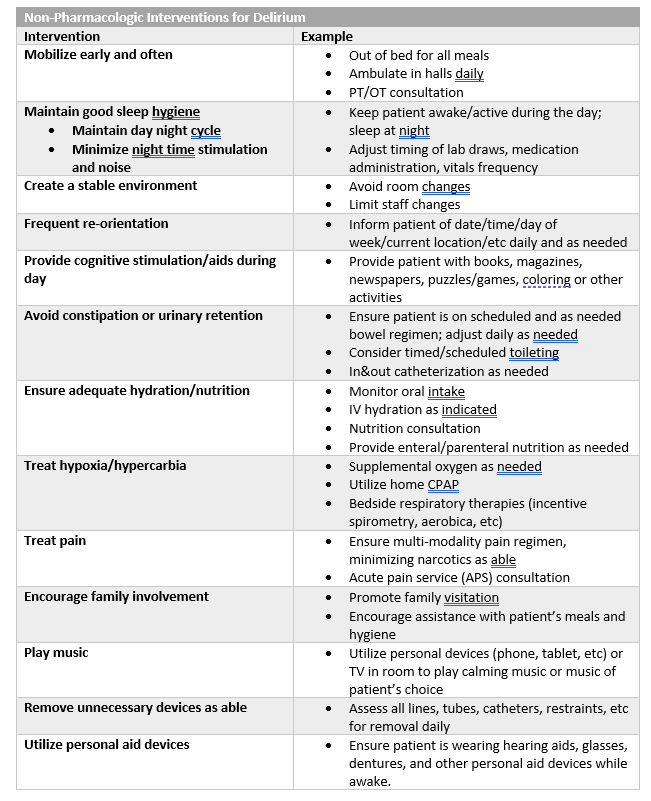
- In addition to correcting/treating contributing factors, provider should also implement delirium treatment interventions beginning with non-pharmacologic interventions first and escalating to pharmacologic interventions as needed. (see FIGURE 4)
-
-
- There are currently no pharmacologic agents shown to prevent the development of delirium or shorten the course of delirium. Pharmacologic agents should be used as needed to treat symptoms of delirium and prevent harm to patient and/or staff.
- If pharmacologic interventions are required, they should be preferentially used on an as needed basis with the minimal dose required to achieve the desired effect (i.e. not agitated or at risk to harm self/staff).
- If scheduled pharmacologic interventions are used, medications should be assessed daily for need and weaned/discontinued once delirium has resolved.
-
-
Follow-up Care
- All patients diagnosed with delirium during his/her hospital stay should be assessed daily and as needed for ongoing signs and symptoms of delirium and potential opportunities to optimize delirium prevention and treatment.
- Diagnosis of delirium should be well documented in the patient’s hospital discharge summary so that post-discharge cognitive and psychiatric sequelae may be assessed at follow-up visits with either trauma providers or patient’s primary care providers.
Outcome Measure and Guideline Adherence
- Diagnosis of delirium is actively tracked through our trauma performance improvement initiatives.
-
-
- Each case will be reviewed by our PI coordinators and TMD/aTMD at a primary and/or secondary review level to confirm diagnosis and assess for opportunities for improvement.
- If opportunities for improvement or trends identified, cases will be further examined at a secondary and/or tertiary review level in our weekly trauma performance improvement conference or monthly multi-disciplinary trauma meetings.
-
-
Related Policies
- Pharmaceutical Management of Post-TBI Neuropsychiatric Symptoms, Acute Care Surgery Patient Pathway, Nebraska Medicine.
Key Contributors
- Emily Cantrell, MD | Division of Acute Care Surgery, Faculty | Principle Author
- Erin Panowicz, APRN | Division of Acute Care Surgery | Author
- Rebecca Sedlak, PharmD, BCCCP | Critical Care Pharmacy, Nebraska Medicine | Author
Last Updated
February, 2024
References
- Williams EC, Estime S, Kuza CM. Delirium in trauma ICUs: a review of incidence, risk factors, outcomes, and management. Curr Opin Anesthesiol. 2023 Apr;36(2):137-146.
- Devlin JW, Skrobik Y, Gelinas C, et al. Clinical practice guidelines for the prevention and management of pain, agitation/sedation, delirium, immobility, and sleep disruption in adult patients in the ICU. Crit Care Med. 2018 Sep;46(9):e825-e873.
- Shoulders BR, Elsabagh S, Tam DJ, et al. Risk factors for delirium and association of antipsychotic use with delirium progression in critically ill trauma patients. Am Surg. 2023 May;89(5):1610-1615.
- Ely EW, et al. Confusion Assessment Method for the Intensive Care Unit. JAMA. 2001; 286:2703-2710.
- Inouye SK, et al. Confusion Assessment Method. Ann Intern Med. 1990; 113:941-948.
Appendix/Supplemental Materials
FIGURE 1--Delirium Screening Tools
(a) Confusion Assessment Method for ICU (CAM-ICU)
(b) Brief Confusion Assessment Method (bCAM) Flow Sheet
FIGURE 2-- Delirium Prevention Strategies
FIGURE 3--Suggested algorithm for management of delirium
FIGURE 4--Non-pharmacologic and pharmacologic interventions for delirium
|
Pharmacologic Interventions for Delirium
|
|||
|
Class/Drug |
Suggested Use |
Dosing |
Adverse Effects |
|
Typical Antipsychotic: Haloperidol (Haldol)
|
Controlling acute severe agitation |
2.5-10 mg (usual dose 5 mg) IV/IM. May repeat Q15min (up to 20 mg) until calm achieved |
Oversedation, QT prolongation, arrythmia, extrapyramidal symptoms, dopaminergic antagonism (avoid in Parkinsons), may lower seizure threshold |
|
Controlling intermittent (or breakthrough) agitation |
2.5- 5 mg Q4H PRN agitation |
||
|
Atypical Antipsychotics: |
|
||
|
Quetiapine (Seroquel) |
Maintaining control of agitation associated with hyperactive/ mixed delirium
|
Typical start: 50 mg PO/perFT Q8-12hr. If effect not achieved at 24 hours, may increase dose (max 400 mg/day). |
Oversedation, QT prolongation (less than IV haloperidol), extrapyramidal symptoms (less than haloperidol) |
|
Olanzapine (Zyprexa) |
Typical start: 5 mg PO/perFT daily. If effect not achieved at 24 hours, may increase dose (max of 20 mg/day). |
||
|
Risperidone (Risperdal) |
Controlling acute agitation |
1-2 mg PO/per FT. May repeat dose in 1-2 hours, up to 6mg in 24 hours.
|
|
|
Central Alpha-2 Agonist: Dexmedetomidine (Precedex) |
Maintaining control of agitation associated with delirium |
If intubated: 0.2-1.5 mcg/kg/hour continuous infusion
If extubated: 0.2-0.7 mcg/kg/hour continuous infusion, order expires at 24-hours, must reassess and reorder if still indicated. |
Restricted to ICU and SDCC. No bolus dosing allowed.
Hypotension, bradycardia, withdrawal (if use prolonged) |
|
Benzodiazepine: Lorazepam (Ativan) |
Controlling severe acute agitation—not typically used as 1st line
|
0.5-1 mg IV/PO/perFT, may repeat in 15 min |
AVOID if able as BZDs causes/exacerbate delirium.
Oversedation |
|
Acceptable option for alcohol withdrawal, agitation in patient with chronic benzo use, agitation in Parkinson’s |
0.25-1 mg IV/PO/perFT Q4-6H PRN agitation |
||
|
Anticonvulsant: Valproic Acid (Depakote) |
For agitation refractory to other agents (ie adequate analgesia/ sedation, antipsychotics). May be especially useful when associated with substance withdrawal or untreated mood (ie bipolar) disorder
|
Typical start: 250 mg IV/PO/perFT Q8H. If effect not achieved at 24 hours, may increase by 250 mg increments.
May use loading dose for acute control: 15 mg/kg (~ 1000 mg)
|
Hepatotoxicity, hyperammonemia, thrombocytopenia, drug interaction with carbapenems
Safe therapeutic range: 50-125 mcg/mL |
|
Endogenous Hormone: Melatonin |
Consider if insomnia is contributing to delirium |
3 mg PO/perFT QHS, may increase to 9 mg |
Daytime drowsiness, limited side effects |
|
SSRI: Trazodone
|
Potentially useful if insomnia is contributing to delirium (2nd line) |
25-50 mg PO/perFT QHS |
Daytime drowsiness, antihistamine effects, sensory distortion, sleep walking |
|
For all added medications for delirium/agitation: · Start at lowest (or a 50% reduced dose) in elderly (ie >65 yoa). · These medications are not for long-term use, reassess daily. Delirium often resolves/improves over several days and the agents should be weaned/discontinued if no longer indicated. |
|||