Whole Blood Usage in Trauma
Purpose:
The implementation of a whole blood program at Nebraska Medicine is designed to optimize our resuscitation practices for the hemorrhaging trauma patient. Whole blood, specifically Low Titer O Whole Blood (LTOWB) immediately delivers a balanced product with less preservative than individual components. It also has the advantage of being more convenient by giving one product instead of three to achieve a balanced transfusion ratio. The goal of this document is to standardize the management of resuscitation with LTOWB for hemorrhagic shock from a traumatic mechanism.
Definitions:
Low Titer O Whole Blood (LTOWB): a complete blood product that contains Type O red blood cells, plasma and platelets. It contains low levels of antibodies making it safe to transfuse to a patient with any blood type. Additionally, LTOWB contains less preservative than its respective components.
Protocol:
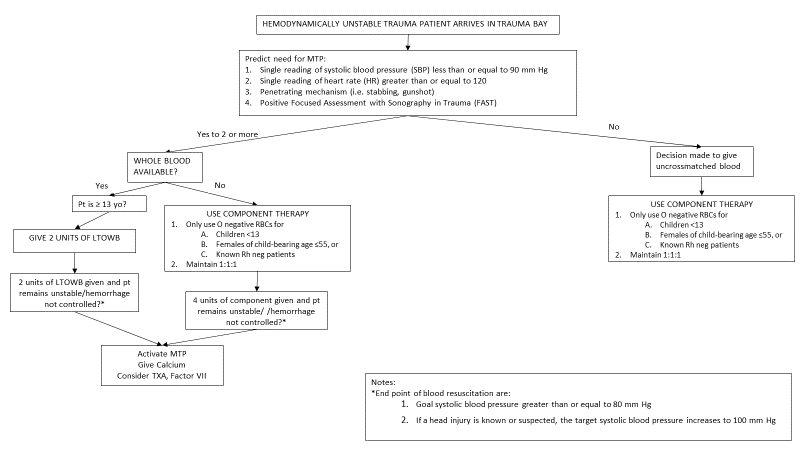
1. This protocol may be initiated in the Trauma Bay in the Emergency Department of Nebraska Medicine for trauma patients only
2. Whole Blood transfusion can only be activated by the attending physician or their surrogate
3. Patients who meet the following criteria are eligible to receive LTOWB
a. Patient is a male or female 13 years of age or older
b. Patient has significant and potentially life-threatening bleeding
c. Anticipated need for massive transfusion protocol (MTP)
4. Once a patient has been deemed eligible and the decision to transfuse LTOWB has been made, the attending physician or their surrogate will direct the ED nurse to begin the transfusion process in accordance with the established MTP/Severe Coagulopathy/Emergency Release Blood Administration Policy (TX 36)
5. After having been deemed eligible to receive LTOWB, a patient may receive a maximum of two units
6. Once a patient receives two units of LTOWB, the MTP should proceed in the usual manner, if indicated
7. Whole blood transfusion initiated in the Trauma Bay may be continued in other patient care areas such as the operating room, interventional radiology, or the intensive care unit
8. All other aspects of transfusion should proceed per established protocols
9. No additional testing is required following administration of LTOWB as the risk of transfusion reaction and hemolysis is thought to be similar to risks associated with uncrossmatched transfusions
FAQ:
Can LTOWB be used for non-traumatic patients?
- No, not at this time. Trauma pilot will run from Nov 2021 to April 2022, then will review for utilization and cost effectiveness.
2. How do I report blood transfused?
- Report LTOWB units separately. For example: 2 u LTOWB, 5 pRBCs, 5 FFP, 1 plts
3. Turn around time after utilization of the 2 u LTOWB in the trauma bay fridge?
- About 2 weeks
4. Do I need additional labs or studies before or after giving LTOWB?
- No, just a T & S within 10 min of accessing the product
5. When does LTOWB expire?
- 3 week topic life after collection, so will be stored in fridge for 2 weeks, then reprocessed
6. Can I give more than 2 units LTOWB?
- No, the 2 units are a bridge to MTP.