Direct Peritoneal Resuscitation
Background, including pathophysiology
The advent of damage control surgery (DCS) has led to a staged approach to the patient in extremis with intra-abdominal hemorrhage and shock that has undoubtedly saved lives. However, massive resuscitation associated with severe hemorrhagic shock involves fluid administration in volumes far in excess of estimated blood loss because of the shift of fluid from the intravascular to the extravascular space. This massive volume load usually results in substantial tissue edema, which can delay abdominal closure.9,10 Acute tissue edema with swelling of the interstitial space secondary to resuscitation is a dominant factor in the inability to close many DCS patients.
Direct peritoneal resuscitation (DPR) is a validated resuscitation strategy in patients with open abdominal wounds. The practice consists of suffusing the peritoneal cavity with a 2.5% glucose-based peritoneal dialysis solution along with traditional intravenous resuscitation. The process leads to microvascular vasodilation and increases visceral and hepatic blood flow1; reverses endothelial cell dysfunction2; improves survival and down regulates the inflammatory response3; reverses established microvascular constriction4; normalizes capillary perfusion density5; and normalizes systemic water compartments6; preserves hepatic blood flow improving survival and preventing hepatic inflammation following hemorrhagic shock7; improves obesity-induced hepatic dysfunction after trauma8; accelerates primary abdominal wall closure after damage control surgery and prevents complications associated with open abdomen9, 10. In addition to observed effects on microcirculation, it has marked ability to decrease visceral edema and normalize body water ratios. In the pediatric population, it has been shown to augment ideal blood flow in necrotizing enterocolitis11. The technique improves inflammation, liver blood flow, and pulmonary edema in brain death12. The technique has been demonstrated safe and effective in humans with no untoward physiologic events or complications9, 10.
Goals of therapy
The goal of Direct Peritoneal Resuscitation is to decrease organ dysfunction, decrease intra-peritoneal inflammatory response, decrease or prevent complications associated with damage control surgery and planned open abdomens and help facilitate early abdominal closure.
The ultimate goal is that patients receiving direct peritoneal resuscitation will achieve significant reduction in time to abdominal closure, decreased number of re-exploration operations as well as higher rate of primary fascial closure.
Indications and Contraindications
Direct Peritoneal Resuscitation is indicated in the treatment of patients with planned open abdomen after damage control surgical procedures where the surgeon is unable to achieve primary fascial closure or in situations where is inadvisable to attempt primary abdominal closure due to planned re-operation, significant peritoneal contamination or inflammation, need to second look due to bowel viability, or other indications. Its use requires the fascia to remain open, with peritoneal contents and fluid controlled ideally with a commercial system such as the Abthera wound vac. However, other temporary abdominal closure strategies are also acceptable as long as they support a system for controlled abdominal fluid affluent.
Contraindications are closed abdominal wall or inability to adequately remove infused peritoneal fluid.
Technique- Equipment and set up with pictures
Operative phase
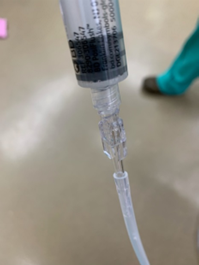
Surgeon performing procedure places drain prior to temporary abdominal closure. Ideally this will involve a standard 19fr round ‘Blake’ type drain placed around the root of the mesentery, though other alternative drains with leur-lock adaptors are acceptable. The Blake drain will have a Medtronic DLP 3mm vascular cannula attached, secured in place with surgical ties (tie several tight silks around the end of the Blake) as needed to ensure watertight connection.
Once in place – the vessel cannula will be capped with a standard IV tubing cap.
Of note- an alternative to the DLP vascular cannula is the following catheter adapter with the Christmas tree on one side and leur lok on the other. These may be more accessible in SICU if for some reason the vascular cannula was not added or available. There were previous reports of leakage but has since been tested and none leak as long as you secure them in there as tight as humanly possible.
ICU
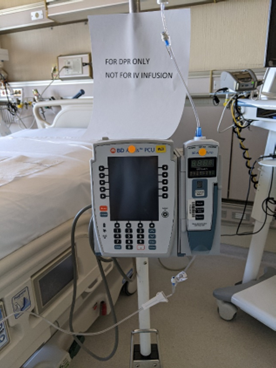
Once the patient arrives in the ICU – the nurse will confirm the order for Direct Peritoenal Resuscitation. A 5000 ml bag Delflex 2.5% peritoneal dialysis solution, will be obtained from pharmacy along with an Alaris IV pump on a separate IV pole and standard Alaris IV tubing. The Alaris IV pump, IV tubing and DPR catheter should be clearly marked “FOR DPR USE, NOT FOR IV INFUSION”.
After ensuring proper function of the temporary abdominal closure device, the DPR IV pump and tubing is connected to the DPR catheter and the infusion initiated at the ordered rate. It should be labeled “DPR only, do not inject” as per the One Chart order.
The infusion is continued until the order is discontinued by the surgical service or the patient is returned to the operative room for abdominal re-exploration. It is acceptable to hook up the Abthera or temporary wound closure to wall suction (at a setting of less than 120 mmHg), but patients should have a KCI VAC unit at bedside as an alternative if needed for fluid removal. Canisters on the temporary abdominal closure device are changed as needed, anticipating that approximately 500cc of fluid will be evacuated from the peritoneal cavity per hour. If using wall suction, the Abthera tubing fits directly into suction tubing and can be secured with tape or tegederm.
Administration/management
ADMINISTRATION:
1. Follow standard steps for nursing procedures.
2. Obtain 5000ml bag of 2.5% Delflex peritoneal dialysis solution from pharmacy.
3. Prime standard Alaris pump IV tubing with solution. For safety, all Direct Peritoneal Dialysis should be infused using an independent Alaris pump separate from all other infusions. Tubing should be labeled to distinguish it from IV infusions and should be reviewed during bedside shift report.
4. To set up, the red frangible pin on the 5L bag should be broken. The bag is connected to IV tubing as usual. The patient should have an abdominal drain in place for infusion. The tubing is connected to the DPR drain using Medtronic 3mm Vascular catheter or other catheter with a leur lock connection. If the connector is not present, the operating physician should be contacted as this is a unique connection device. Insert the “christmas tree” end of the connector into the Blake drainand connect the pump tubing using the leur lock end.
5. Program pump according to administration instructions in ACP orders. RN should expect orders for 500ml bolus, then continue infusion at a rate of 1.5ml/kg/hr until AbThera would vac is removed or orders are discontinued. A new bag of Delflex solution should be used every 24 hours while resuscitation is ongoing.
6. The dialysate must be on a separate pole and pump.
7. Label the Blake drain most distal from the site indicating the type of drain.
8. Label the fluid tubing most distal from the bag with type of fluid.
9. The solution will automatically circulate in the peritoneal cavity and be evacuated through the negative pressure AbThera wound vacuum, either to the VAC canister or wall suction <120 mmHg.
10. DPR solution infused should be recorded as “input” by the Registered Nurse in the patient’s EHR in the row for the Abdominal Drain DPR and wound vac output should be included as “output” in the medical record under the row included with the LDA for the incision.
11. Change wound vac containers as needed. STOP the DPR infusion anytime the wound vac is not running/functioning for more than a short period of time.
12. NEVER clamp the wound vac tubing while the infusions are running. If you need to trouble shoot the wound vac, STOP the DPR infusion pump until wound vac is functioning. Contact the surgical team for assistance as needed.
Ordering/order sets
See flier for order set details. Please note the rates of infusion for the initial bolus and maintenance rates should not be adjusted.
Ins and outs will be recorded as shown in the flier also.
Complications and concerns
Inadequate removal of peritoneal fluid leading to potential increased intra-abdominal pressure, intra-abdominal hypertension and potential abdominal compartment syndrome.
Leakage of dialysate from instillation or evacuation process due to improper/inadequate set-up.
If either of the above is a concern, stop the infusion, call the provider. Do not stop the suction on the VAC or wall.
Abby Josef, MD
References
1. Hurt RT, Zakaria el R, Matheson PJ, Cobb ME, Parker JR, Garrison RN. Hemorrhage-induced hepatic injury and hypoperfusion can be prevented by direct peritoneal resuscitation. J Gastrointest Surg. 2009;13(4):587-94.
2. Zakaria el R, Li N, Garrison RN. Mechanisms of direct peritoneal resuscitation-mediated splanchnic hyperperfusion following hemorrhagic shock. Shock. 2007;27(4):436-42.
3. Garrison RN, Conn AA, Harris PD, Zakaria el R. Direct peritoneal resuscitation as adjunct to conventional resuscitation from hemorrhagic shock: a better outcome. Surgery. 2004;136(4):900-8.
4. Zakaria el R, Garrison RN, Kawabe T, Harris PD. Direct peritoneal resuscitation from hemorrhagic shock: effect of time delay in therapy initiation. J Trauma. 2005;58(3):499-506; discussion -8.
5. Zakaria el R, Li N, Matheson PJ, Garrison RN. Cellular edema regulates tissue capillary perfusion after hemorrhage resuscitation. Surgery. 2007;142(4):487-96; discussion 96 e1-2.
6. Zakaria el R, Matheson PJ, Flessner MF, Garrison RN. Hemorrhagic shock and resuscitation mediated tissue water distribution is normalized by adjunctive peritoneal resuscitation. J Am Coll Surg. 2008;206(5):970-80; discussion 80-3.
7. Hurt RT, Matheson PJ, Smith JW, Zakaria el R, Shaheen SP, McClain CJ, Garrison RN. Preservation of hepatic blood flow by direct peritoneal resuscitation improves survival and prevents hepatic inflammation following hemorrhagic shock. Am J Physiol Gastrointest Liver Physiol. 2012;303(10):G1144-52.
8. Matheson PJ, Franklin GA, Hurt RT, Downard CD, Smith JW, Garrison RN. Direct peritoneal resuscitation improves obesity-induced hepatic dysfunction after trauma. J Am Coll Surg. 2012;214(4):517-28; discussion 28-30.
9. Smith JW, Garrison RN, Matheson PJ, Franklin GA, Harbrecht BG, Richardson JD. Direct peritoneal resuscitation accelerates primary abdominal wall closure after damage control surgery. J Am Coll Surg. 2010;210(5):658-64, 64-7.
10. Smith JW, Neal Garrison R, Matheson PJ, Harbrecht BG, Benns MV, Franklin GA, Miller KR, Bozeman MC, David Richardson J. Adjunctive treatment of abdominal catastrophes and sepsis with direct peritoneal resuscitation: indications for use in acute care surgery. J Trauma Acute Care Surg. 2014;77(3):393-8; discussion 8-9.
11. Maki AC, Matheson PJ, Shepherd JA, Garrison RN, Downard CD. Intestinal microcirculatory flow alterations in necrotizing enterocolitis are improved by direct peritoneal resuscitation. Am Surg. 2012;78(7):803-7.
12. Smith JW, Ghazi CA, Cain BC, Hurt RT, Garrison RN, Matheson PJ. Direct peritoneal resuscitation improves inflammation, liver blood flow, and pulmonary edema in a rat model of acute brain death. J Am Coll Surg. 2014;219(1):79-87.
13. Walker SK, Matheson PJ, Schreiner MT, Smith JW, Garrison RN, Downard CD. Intraperitoneal 1.5% Delflex improves intestinal blood flow in necrotizing enterocolitis. J Surg Res. 2013;184(1):358-64.
14. Downard CD, Matheson PJ, Shepherd JA, Maki AC, Garrison RN. Direct peritoneal resuscitation augments ileal blood flow in necrotizing enterocolitis via a novel mechanism. J Pediatr Surg. 2012;47(6):1128-34.
15. Matheson PJ, Mays CJ, Hurt RT, Zakaria ER, Richardson JD, Garrison RN. Modulation of mesenteric lymph flow and composition by direct peritoneal resuscitation from hemorrhagic shock. Arch Surg. 2009;144(7):625-34.
16. Matheson PJ, Hurt RT, Franklin GA, McClain CJ, Garrison RN. Obesity-induced hepatic hypoperfusion primes for hepatic dysfunction after resuscitated hemorrhagic shock. Surgery. 2009;146(4):739-47; discussion 47-8.
17. Zakaria el R, Tsakadze NL, Garrison RN. Hypertonic saline resuscitation improves intestinal microcirculation in a rat model of hemorrhagic shock. Surgery. 2006;140(4):579-87; discussion 87-8.
18. Cryer HM, Gosche J, Harbrecht J, Anigian G, Garrison N. The effect of hypertonic saline resuscitation on responses to severe hemorrhagic shock by the skeletal muscle, intestinal, and renal microcirculation systems: seeing is believing. Am J Surg. 2005;190(2):305-13.
19. Zakaria el R, Garrison RN, Spain DA, Harris PD. Impairment of endothelium-dependent dilation response after resuscitation from hemorrhagic shock involved postreceptor mechanisms. Shock. 2004;21(2):175-81.
20. Zakaria el R, Hurt RT, Matheson PJ, Garrison RN. A novel method of peritoneal resuscitation improves organ perfusion after hemorrhagic shock. Am J Surg. 2003;186(5):443-8.
21. Zakaria el R, Garrison RN, Spain DA, Matheson PJ, Harris PD, Richardson JD. Intraperitoneal resuscitation improves intestinal blood flow following hemorrhagic shock. Ann Surg. 2003;237(5):704-11; discussion 11-3.
22. Weaver JL, Smith, JW. (2016). Direct Peritoneal Resuscitation: A review. International Journal of Surgery 2016; 33:237-241.